“Same Subject Matter” in Priority Seen from the Second Instance Judgment of “Takeda” Case
Under the local practice, the criteria for priority verification are often regarded as being similar to those for “novelty” evaluation or “new-matter introduction” determination. Recently, the Supreme People's Court issued an administrative judgment of second instance for the case of invalidation against patent No. CN201210332314.2 entitled “dipeptidyl peptidase inhibitors for treating diabetes” (hereinafter referred to as “Takeda case”). In the following, the criteria for determining “the same subject matter” in priority verification will be studied from this administrative judgment.
The Chinese patent law provides in Article 29 the provisions for both foreign and domestic priorities, as follows:
“Where, within twelve months from the date on which any applicant first filed in a foreign country an application for a Patent for invention or utility model, or within six months from the date on which any applicant first filed in a foreign country an application for a patent for design, he or it files in China an application for a patent for the same subject matter, he or it may, in accordance with any agreement concluded between the said foreign country and China, or in accordance with any international treaty to which both countries are party, or on the basis of the principle of mutual recognition of the right of priority, enjoy a right of priority.
“Where, within twelve months from the date on which any applicant first filed in China an application for a patent for invention or utility model, he or it files with the Patent Administration Department under the State Council an application for a patent for the same subject matter, he or it may enjoy a right of priority.”
It is known from the above provisions that, if a subsequent application is eligible for priority of an earlier application, at least the following conditions shall be satisfied: (1) the subsequent application shall be filed within twelve months (for an invention or utility model) or six months (for a design) from the filing date of the earlier application, (2) the earlier application shall be a “first application”, and (3) the subsequent application shall have the “same subject matter” as the earlier application.
As to how to determine whether the subsequent application have the “same subject matter” as the earlier application, “Patent Examination Guidelines 2023” gives the following definition in Part II, Chapter 3, Section 4.1.2: “the inventions or utility models with the same subject matter as defined in Article 29 of the Patent Law refer to the inventions or utility models that relate to the same technical field, and have the same the technical problem to be solved, the technical solution and the expected technical effects', the so-called “four-element” principle. This might be the reason why the criteria for priority verification are often regarded as being similar to those for “novelty” evaluation.
“Patent Examination Guidelines 2023” further states in Part II, Chapter 8, Section 4.6.2 that, for the verification of the “same subject matter” between the earlier application and the subsequent application, it is necessary to “determine whether the technical solutions of the claims of the subsequent application are clearly recited in the earlier application (including the description and the claims, but not the abstract)”. As to the wording “clearly recited”, it does not require complete consistency in the literal expression, but requires that the technical solutions defined by the claims of the subsequent application are clearly described in the earlier application; however, if the earlier application only provides a general or vague description of one or some of the technical features of the above technical solutions, or even only provides a hint thereof, while the subsequent application claiming priority adds detailed description for this or these technical features, so that those skilled in the art believe that the technical solutions cannot be directly and unambiguously obtained from the earlier application, the earlier application cannot serve as the basis for the subsequent application to claim priority. The expression of “directly and unambiguously obtained” as used might be the reason why the criteria for priority verification are often regarded as being similar to those for “new matter introduction” determination.
The “Takeda case” has clarified the criteria for determining the same subject matter in priority verification.
The patent in dispute relates to dipeptidyl peptidase inhibitors for treating diabetes, and its granted claim 1 reads:
“Use of compound I in the manufacture of a pharmaceutical composition for treating type II diabetes by administering orally compound I in a daily dosage of 5 mg to 250 mg, wherein compound I has a structure of formula:
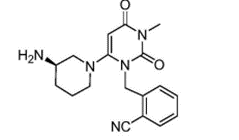
and is present in a form of a pharmaceutically acceptable salt or freebase.”
The patent in dispute claims priorities of US60/717,558 (filed on September 14, 2005) and US60/747,273 (filed on May 15, 2006).
When filing a request for invalidation against the patent in dispute, the petitioner submitted a copy of the patent publication No. WO2005/095381A1 (hereinafter referred to as “evidence 4”) entitled “Dipeptidyl Peptidase Inhibitor” and filed on December 15, 2004, and indicated that evidence 4 discloses all the technical features of claim 1 of the patent in dispute, except the technical feature of “daily dosage of 5 mg to 250 mg”, and that the daily oral dosage of a compound is a feature in relation to administration, which is only reflected in doctor's medication process and has no limitation effect on the pharmaceutical process, and thus does not have any substantive effect on the protection scope of claim 1. As a result, evidence 4 has disclosed the same subject matter as claim 1 of the patent in dispute, which makes neither of the two earlier applications to which the patent in dispute claims priorities be the “first application”. Therefore, claim 1 of the patent in dispute is not entitled to enjoy priorities of the two earlier applications.
In “Takeda case”, the dispute focus on whether the patent in dispute is entitled to enjoy priorities of the two earlier applications, i.e., whether evidence 4 has disclosed the same subject matter as the patent in dispute. More particularly, the dispute lies in whether the technical feature of “a daily dosage of 5 mg to 250 mg” has a limitation effect in the priority verification.
During the examination of the request for invalidation and during the subsequent first trail, both the CNIPA and the Court of First Instance hold that the technical feature of “daily dosage” does not have a limitation effect on the medical use of a substance, claim 1 defined by such technical feature does not relate to a subject matter different from that of evidence 4, and therefore the patent in dispute is not entitled to enjoy priorities of the two earlier applications.
However, during the second trial of this case, the Supreme Court holds that, in the priority verification, the entire content as defined in the claims, including the technical feature of “daily dosage of 5 mg to 250 mg”, should be considered, and evidence 4 cannot constitute the first application having the same subject matter as the patent in dispute, as it does not disclose such technical feature.
It can be seen from the above that the Supreme Court made it clear for the first time that, when determining whether the subsequent and earlier applications have “the same subject matter” in priority verification, all the technical features of the technical solutions of the claims of the subsequent application shall be taken into consideration, with “directly and unambiguously obtained” as the criterion. Secondly, the Supreme Court also makes it clear that the priority verification is only for the purpose of determining whether the filing date of the earlier application can be regarded as the priority date, and whether the filing date of the earlier application can be regarded as the filling date of the subsequent application according to the provisions of the patent law; and the priority verification does not involve examination on patentability, and thus shall not introduce consideration for patentability examination. In other words, the priority verification is a prerequisite for the novelty evaluation, and has different criteria for determining “the same subject matter” from that for novelty evaluation, and thus the criteria for novelty evaluation shall not be introduced into priority verification.
The judgment not only clarifies the criteria for determining “the same subject matter” in priority verification, but also gives the author a new understanding of the amendment to the claims related to a method for diagnosing or treating a disease. A foreign patent application may include one or more claims related to a method for diagnosing or treating a disease, which are patentable under its local patent practice. Such claims may include a lot of technical features such as administration subject, dosage, time interval and the like, which, under the patent practice in China, might be deemed as having no substantive effect on the pharmaceutical process but being only embodied in the medication process. When filing a patent application in China based on such foreign patent application via the Paris Convention or the PCT route, the applicant may consider redrafting such claims into Swiss-type claims via voluntary amendments or in response to the examiner’s comments, so as to overcome the defect of lacking unpatentable subject matter. Based on the teachings of this case, when redrafting such claims into Swiss-type claims, it is preferable to include such technical features in the claims as much as possible. Although such technical features per se might not confer novelty to the Swiss-type claims including the same for they have no substantive effect on the pharmaceutical process, they might make the subsequent application eligible for priority of the earlier application in priority verification, so as to take the filing date of the earlier application as the date for determining the prior art, which is quite important in evaluating novelty and inventiveness of the subsequent application. Therefore, when redrafting the claims related to a method for diagnosing or treating a disease, it is not advisable to delete one or more technical features which are regarded as having no substantive effect on novelty and inventiveness evaluation, as deletion of such technical features might render the patent ineligible for priority and result in prejudice to the interests of the patentee.